China Viral Transport Medium Tube: Swabs & VTM Supplier
5777The 10-mix-1 virus transport medium has become an effective tool for covid-19 PCR assays because it improves the efficiency of sampling and detecti...
View detailsSearch the whole station Lab Supply
When a lab reports a false negative, the blame rarely falls on the collection device. Yet, more often than not, that’s exactly where the error begins.
In the world of molecular diagnostics, the journey from patient to result is a fragile one. We obsess over PCR machines, reagents, and thermocyclers, but the humble viral transport medium (VTM)—the tube that cradles the sample from swab to analysis—is frequently treated as an afterthought. It shouldn’t be.
The truth is, a high-performance VTM system is not just a container. It is the first and most critical gatekeeper of diagnostic accuracy. While most buyers focus on price and delivery time, a handful of unspoken criteria separate the truly reliable systems from the rest. Here is what defines excellence—and how to spot it.
In a global market flooded with unbranded and unverified collection kits, regulatory clearance is not just paperwork; it is the ultimate stress test.
True high-performance VTMs don’t just claim quality—they prove it through the world’s most stringent regulatory frameworks. Achieving China NMPA, US FDA 510(k) clearance, and EU CE MDR certification simultaneously is remarkably rare. Each pathway demands rigorous validation: stability studies, biocompatibility tests, and clinical performance data that most manufacturers simply cannot produce.
In the complex global regulatory landscape, only a handful of manufacturers hold all three credentials. HCY Technology is one of them. For laboratories and procurement teams, this triple lock isn’t just a badge of honor—it’s the elimination of compliance risk before a single sample is collected.
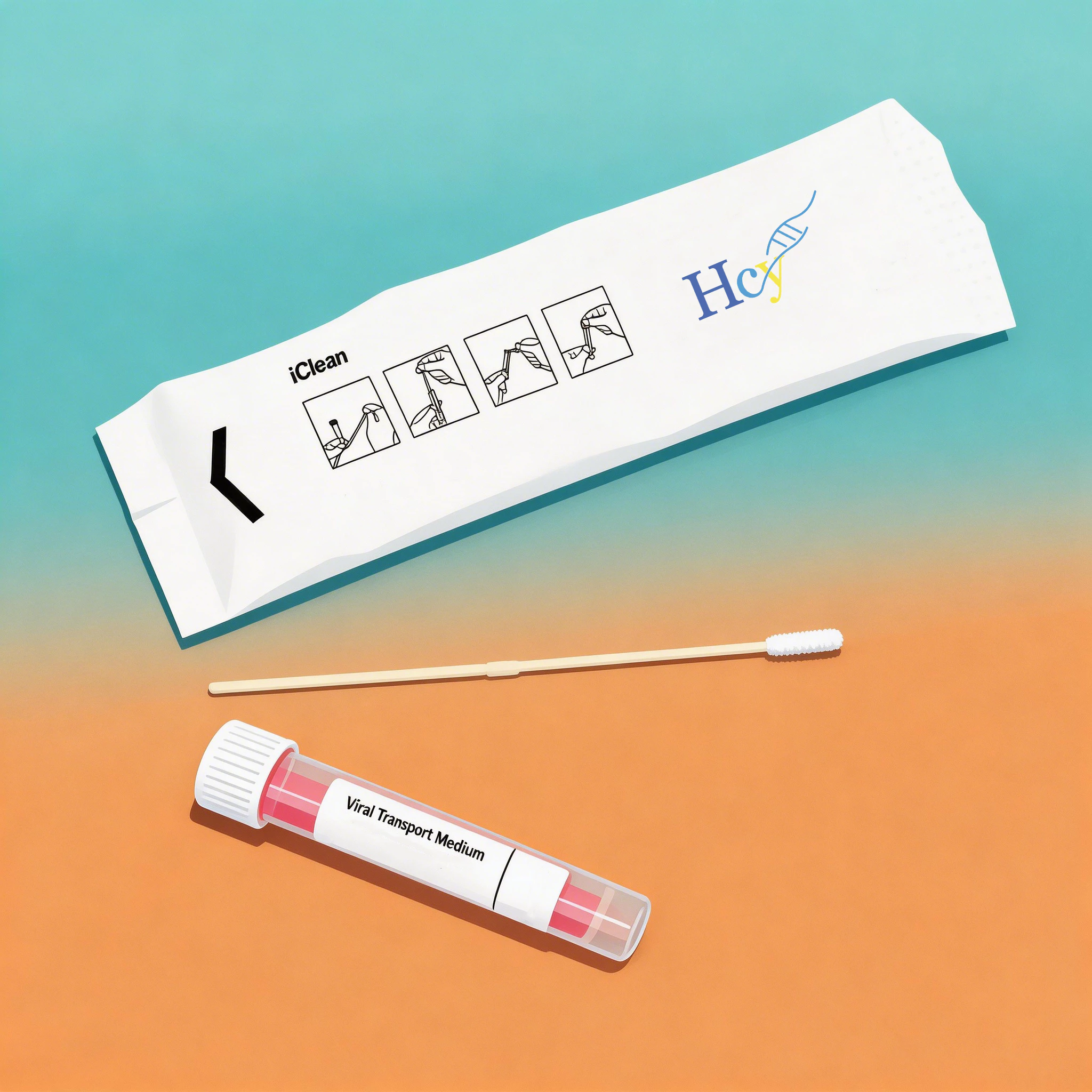
Not all VTMs are created equal—and the choice between non-inactivating and inactivating formulations is just the beginning. Performance is measured in hours, degrees, and recovery rates.
A truly high-performance VTM does two things exceptionally well. First, it maintains viral integrity during transport. For non-inactivating formulations, this means preserving the virus’s structural integrity for downstream applications like viral culture. Second, it keeps bacterial and fungal contaminants in check, preventing overgrowth that can trigger false positives or degrade RNA.
The numbers matter here. In validated systems, viral RNA remains stable at 2–8°C for over 48 hours, with recovery rates exceeding 90% for common respiratory viruses such as influenza A and B. Beyond stability, the medium’s composition—balanced salt solutions, protein stabilizers, and precise antibiotic concentrations—determines whether a sample arrives at the lab intact or compromised.
HCY’s patented VTM formulation has been refined specifically to meet the demands of both PCR diagnostics and viral culture workflows—a combination that many laboratories consider the gold standard when evaluating VTM performance.
The swab itself is often underestimated. Yet, in viral transport, the surface area and release efficiency of the swab directly impact whether a sample yields a detectable result.
Conventional cotton or rayon swabs trap cells within their fibers, releasing only a fraction of the collected material into the transport medium. Flocked swabs, by contrast, are engineered with short, perpendicular fibers that act like a micro-brush. They capture cells across their entire surface and—crucially—release them efficiently into the medium through capillary action.
This is not a minor detail. Studies consistently show that flocked swabs deliver significantly higher cell yields compared to traditional alternatives. For low-viral-load patients, that difference can mean the line between detection and a missed diagnosis.
High-performance viral transport is not the result of a single feature. It is the convergence of three elements: regulatory rigor that proves reliability, a formulation that preserves or stabilizes with precision, and a swab design engineered for maximum yield.
These are the unspoken criteria—the factors that separate products built for diagnostics from those simply filling a catalog. When a VTM system quietly meets all three, it does something remarkable: it removes uncertainty from the very first step.
And in diagnostics, removing uncertainty is what excellence looks like.
The 10-mix-1 virus transport medium has become an effective tool for covid-19 PCR assays because it improves the efficiency of sampling and detecti...
View detailsVTM allows the safe transfer of viruses, chlamydiae, and mycoplasma for further research and analytics. The special formulation ensures the best possible recovery of the samples and the addi...
View detailsDisposable virus transport medium can help medical staff collect viruses and monitor samples.
View detailsLooking for a universal VTM for flu, RSV, and COVID-19? HCY’s viral transport medium offers >70% recovery, scalable supply, and lab‑ready design.
View detailsWe value your privacy We use cookies to enhance your browsing experience, serve personalized ads or content, and analyze our traffic. By clicking "Accept All", you consent to our use of cookies.
Our Privacy Policy