China Viral Transport Medium Tube: Swabs & VTM Supplier
5749The 10-mix-1 virus transport medium has become an effective tool for covid-19 PCR assays because it improves the efficiency of sampling and detecti...
View detailsSearch the whole station Lab Supply
Virus samples are usually not tested immediately after collection; the collected samples are stored and transported. Since the virus itself will quickly lyse in vitro and thus affect the results of subsequent testing, virus transport media are required to preserve and transport the virus.
Virus samples are usually not tested immediately after collection; rather, the collected samples are stored and transported. Since the virus itself will quickly lyse in vitro and thus affect the results of subsequent testing, virus transport media are required to preserve and transport the virus.
Inactivated virus preservation solution is composed of a virus lysis type preservation solution modified from nucleic acid extraction lysate. It effectively prevents secondary infection by operators and contains inhibitors that protect the viral nucleic acid from degradation, thus enabling subsequent detection by NT-PCR. The inactivated virus transport medium allows viruses to be stored at room temperature for longer, saving the cost of preserving and transporting viral samples.
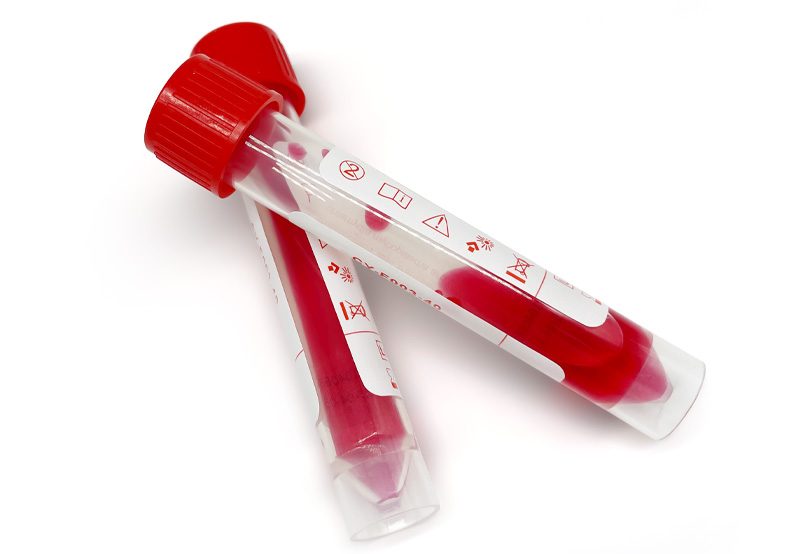
Non-inactivated virus preservation solution is mainly a modified virus maintenance fluid type preservation solution based on the transport medium. It can maintain the in vitro activity of the virus and the integrity of antigen and nucleic acid and contains Hanks base solution and other antibiotics to protect the virus’s protein shell from decomposition and maintain the originality of virus samples to a greater extent. In addition to nucleic acid extraction and testing, non-inactivated virus transport media can also be used for virus culture, isolation, and antigen detection but requires low-temperature storage if it is to be stored for a long time after sampling.
“HCY, Health care for you” is our forever mission. We dedicate to offering safe & reliable products and medical services with our global creditable partners. HCY has already supplied to WHO, MAYO clinic, MGI, DDC, Yale University, Qorvo, Quanterix, Thomas Scientific, SD biosensor, Cardinal Health, Cleveland Clinic, Mars Petcare & LumiraDx, etc. in the past years.
The 10-mix-1 virus transport medium has become an effective tool for covid-19 PCR assays because it improves the efficiency of sampling and detecti...
View detailsVTM vs. saline for COVID testing: New study shows surprising results. Discover if expensive viral transport media is really necessary.
View detailsDiscover the unspoken criteria for a high-performance viral transport medium: regulatory triple lock, formulation stability, and flocked swab desig...
View detailsViral Transport Media (VTM) is a specialized, buffered solution designed to preserve the integrity, viability (or nucleic acid) of viral specimens during collection, transport, and short-term stor...
View detailsWe value your privacy We use cookies to enhance your browsing experience, serve personalized ads or content, and analyze our traffic. By clicking "Accept All", you consent to our use of cookies.
Our Privacy Policy